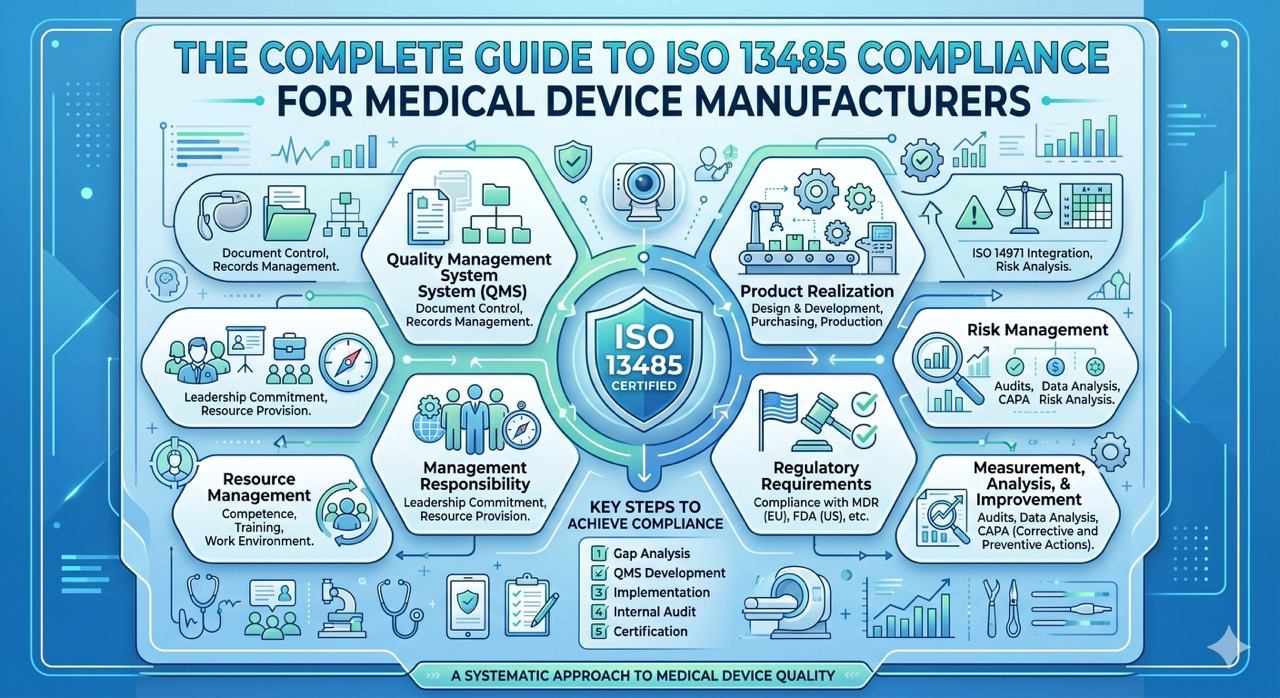
As manufacturing organizations continue to face increasingly stringent regulatory requirements, the adoption of Application Lifecycle Management (ALM) becomes a critical consideration. ALM is a comprehensive approach to managing the entire software development process from start to finish, including requirements management, design, coding, testing, deployment, and maintenance. But when is the right time to implement ALM in a regulated manufacturing organization?
The only ALM and QMS solution
While there is no one-size-fits-all answer to this question, there are several factors that organizations should consider when deciding whether to implement ALM. Here are five key factors to consider:
- Regulatory Compliance: Compliance with regulatory requirements, such as those imposed by the FDA, is a crucial consideration for manufacturing organizations. Implementing ALM can help to ensure that regulatory requirements are met, and that all necessary documentation is in place.
- Complexity of Products: As products become more complex, the need for a more comprehensive approach to software development becomes more critical. ALM provides a holistic framework for managing the development of complex products, ensuring that all aspects of the development process are tracked and managed.
- Increased Collaboration: As organizations grow, collaboration becomes more critical, particularly in a regulated environment. ALM provides a collaborative platform that enables teams to work together effectively, ensuring that all stakeholders are on the same page.
- Cost of Quality: Quality control and assurance are critical in regulated manufacturing, and the cost of quality can be significant. ALM helps to minimize the cost of quality by identifying issues early in the development process, reducing the likelihood of costly rework or recalls.
- Scalability: As organizations grow, they need to be able to scale their software development processes to keep up with demand. ALM provides a scalable framework for managing the entire software development process, ensuring that development processes can be scaled to meet the needs of the organization.

When considering these factors, it becomes clear that implementing ALM in a regulated manufacturing organization can provide significant benefits. However, it is also essential to consider the timing of the implementation. Here are some considerations:
- Organizational Maturity: Organizations should have a certain level of maturity to adopt ALM, with a solid foundation in place in terms of existing development processes, tools, and culture.
- Resource Allocation: Implementing ALM requires a significant investment in terms of time, effort, and resources. Organizations must ensure that they have sufficient resources available to support the implementation process.
- Business Case: There should be a clear business case for implementing ALM, with a well-defined set of benefits that can be realized through the adoption of this approach.
- Change Management: Implementing ALM will involve significant changes to existing development processes, tools, and culture. Organizations must have a well-defined change management plan in place to ensure that the implementation is successful.
- Training: ALM requires a significant level of knowledge and expertise, both in terms of the toolset and the underlying development processes. Organizations must provide adequate training and support to ensure that staff members are equipped to use ALM effectively.

In addition to the five factors outlined above, it is also important to consider the role of software validity and compliance to the 21 CFR Part 11 standard requirement when implementing ALM in a regulated manufacturing organization.
21 CFR Part 11 is a set of regulations issued by the US Food and Drug Administration (FDA) that establishes requirements for electronic records and electronic signatures used in FDA-regulated environments. The regulation applies to all electronic records, including those that are created, modified, maintained, archived, retrieved, or transmitted.
When implementing ALM in a regulated manufacturing organization, compliance with 21 CFR Part 11 is crucial, as it ensures the validity and reliability of electronic records and signatures. Failure to comply with these regulations can result in costly recalls, fines, and damage to the organization’s reputation.
Implementing ALM can help organizations to comply with 21 CFR Part 11 by providing a comprehensive platform for managing the entire software development process, from requirements management to testing and deployment. ALM tools can help to ensure that electronic records are accurate, complete, and reliable, and that electronic signatures are secure and traceable.
By implementing ALM, organizations can also automate many of the manual processes associated with recordkeeping, reducing the risk of errors and ensuring that all necessary documentation is in place. This can help to streamline compliance efforts, reducing the risk of costly recalls and other regulatory issues.
In conclusion, implementing ALM in a regulated manufacturing organization can provide significant benefits in terms of compliance, collaboration, quality, scalability, and complexity management. However, organizations must carefully consider the timing of the implementation and ensure that they have the necessary resources, business case, change management plan, and training in place to support the implementation process. With careful planning and execution, ALM can help organizations to manage the entire software development process more effectively, ensuring that regulatory requirements are met, and products are delivered to market with a high degree of quality and efficiency.